Roundup® (a glyphosate-based herbicide) leads to neurotoxicity in hippocampus of immature rats
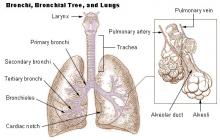
The aim of this study was to investigate whether Roundup® (a glyphosate-based herbicide) leads to neurotoxicity in hippocampus of immature rats following acute (30min) and chronic (pregnancy and lactation) pesticide exposure. Maternal exposure to pesticide was undertaken by treating dams orally with 1% Roundup® (0.38% glyphosate) during pregnancy and lactation (till 15-day-old). Hippocampal slices from 15 day old rats were acutely exposed to Roundup® (0.00005-0.1%) during 30min and experiments were carried out to determine whether glyphosate affects 45Ca2+ influx and cell viability. Moreover, we investigated the pesticide effects on oxidative stress parameters, 14C-α-methyl-amino-isobutyric acid (14C-MeAIB) accumulation, as well as glutamate uptake, release and metabolism. Results showed that acute exposure to Roundup® (30min) increases 45Ca2+ influx by activating NMDA receptors and voltage-dependent Ca2+ channels, leading to oxidative stress and neural cell death.